Revolutionizing Biology with Nanocrystal Molecules As Probes
Cells orchestrate dynamically cooperative operations of a variety of biomolecular machineries to tightly regulate key biological functions such as signaling, transport, replication, and transcription. In order to investigate such highly complicated dynamic events of biomolecular machineries in living objects; our lab develops advanced nanoprobing systems, termed nanocrystal Molecules As Probes (nanoMAPs.) The nanoMAPs are constructed by assembling of multiple nanocrystal components in which individual nanocrystal components (i.e. nanocrystal atoms) are either electronically, magnetically, optically, or mechanically coupled to each other. The chemical and physical properties of the nanoMAPs are a function of interparticle couplings, and hence a function of the interparticle distance, the bonding mode, and the nanocrystal molecule geometry (e.g. symmetry). Unlike conventional single molecule- or single nanocrystal-based bioprobes that only report the spatial distribution of target bio-molecules, such unique coupling phenomena of the nanoMAPs provide optical and magnetic transduction of dynamic structural and environmental changes across single biomolecule. We expect that the development of new nanoMAPs will enable deeper understanding of the fundamental dynamics of bio-molecular machineries in living objects. Specifically, we develop a set of new nanoMAP systems that include magnetic, plasmonic, and fluorescent nanoMAPs to investigate structures, forces, interaction, and reaction of key biomolecules related to cell-to-cell communication (Notch), angiogenesis, gene regulation, and neuron signaling.
Research
Single molecule study of membrane receptor tyrosine kinase dynamics
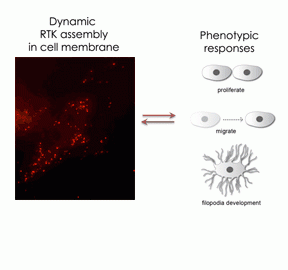
Membrane receptor tyrosine kinases (RTKs) are key regulators of cellular processes and also known to have a critical role in the cancer development and progression. Most RTKs exist as a free monomeric sub-unit, but target ligand binding such as polypeptides and hormones to their extra cellular domain promotes RTK dimerization and even oligomerization. The membrane association of the RTKs then activates intracellular signal cascades related to key cell developments such as proliferation and differentiation. Interestingly, even in the case of genetically identical cells, each cell expresses different frequency and spatial distribution of the RTK assembly and hence behaves differently and sometimes such inhomogeneity of RTK assemblies directs distinctly different fates of each cell. To understand how the dynamic assembly of RTKs determines the phenotypic responses of individual cells, we develop a new nanoMAP based single molecule imaging technique and, by using this new technique, we study real-time RTK dynamics in live cell membranes.
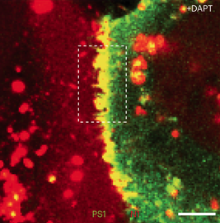
Individually in apoptotic cells: In situ single molecule study using plasmonic nanoMAPs
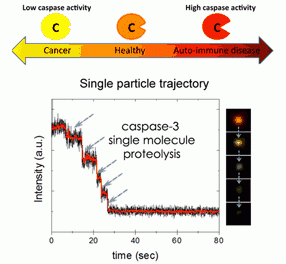
Apoptosis is a well-choreographed mode of cell death where caspases (cysteine-aspartic acid proteases) take center stage in orchestrating cellular demise. Upon commencement of the caspase activation cascades, hundreds of protein substrates are cleaved and the hallmarks of apoptosis (nuclear condensation, cell shrinkage, membrane blebbing, and DNA fragmentation) begin to take their form. Despite remarkable advances in biochemical characterization of apoptotic lysates, the individualistic behavior of cells in a dying population is lost by synchronization of ensemble analyses. It has been postulated that the trajectory of cell death occurs on the order of hours or days, where once a certain internal signaling threshold has been met the cell follows an irreversible death sentence where it is doomed to die within 10 minutes. This paradigm could only have been brought to light with extensive single molecule and cell analysis. By using plasmonic nanoMAPs that show characteristic optical signals against the apoptotic signal progression, we perform single molecule monitoring of cell to provide signal progression, we perform single molecule monitoring of cell to provide key quantitative hallmarks to help describe apoptosis in diverse cell types, cancerous or otherwise, that are prominently mentioned throughout the cell death literature. Such questions call for the construction of sensitive imaging tools to continuously monitor processes at single molecule resolution that transpire along time courses unanticipated by the observer.
Single molecule kinetic study of helicases using nanoMAP protractors
Helicase is an essential motor protein for nucleic acid replication and transcription. Despite recent progress in understanding the mechanism of the molecular motor, development of high-resolution single molecule imaging technique is expected to provide deeper insights of undiscovered molecular mechanisms and kinetic processes of helicases. To investigate molecular mechanisms of helicase activity, we develop a new nanoMAP plasmon protractor technique having single-base pair resolution. Unlike conventional single molecule techniques that only provide distance-displacement of target nucleic acids, the pIasmonic protractor allows us to directly translate the rotational motion of the nucleic acid caused by the helicase to optical signals. This new technique has the potential to provide extremely high sensitivity and spatial resolution enough to detect single base pair melting.
Publications
- Cheryl Tajon, Daeha Seo, Jennifer Asmussen, Neil Shah, Young-wook Jun* & Charles S. Craik* "Sensitive and selective plasmon ruler nanosensors for monitoring the apoptotic drug response in leukemia" submitted.
- Daeha Seo, Justin Farlow, Kade Southard, Young-wook Jun* & Zev J. Gartner* "Production & targeting of monovalent quantum dots" The Journal of Visualized Experiments (JOVE), in press.
- Cheryl Tajon, Young-wook Jun & Charles S. Craik* "Single-molecule sensing of caspase activation in live cells via plasmon coupling nanotechnology" Methods in Enzymology, in press.
- Jennifer Gajan, Elisabeth A. Lasaster, Cheryl Tajon, Juan Ose-Proetp, Young-wook Jun, Barry S.Taylor, Alma Burlingame, Charles S. Craik & Neil P. Shah* "MEK-dependent negative feedback underlies BCR-ABL mediated oncogene addiction" Cancer Discovery, 4, 200 (2014).
- Justin Farlow, Daeha Seo, Kyle E. Broaders, Marcus Taylor, Zev J. Gartner* & Young-wook Jun* "Formation of targeted monovalent quantum dots by steric exclusion" Nature Methods, 10, 1203 (2013).
Before UCSF
- Young-wook Jun, Hea-Won Chung, Jung-tak Jang & Jinwoo Cheon* “Multiple twinning drives nanoscale hyper-branching of titanium dioxide nanocrystals" Journal of Materials Chemistry 21, 10283 (2011).
- Sassan Sheikholeslami‡, Young-wook Jun‡, Prashant Jain‡ & A. Paul Alivisatos* “Coupling of optical resonances in an compositionally asymmetric plasmonic nanoparticle dimer” Nano Letters 10, 2655 (2010).
- Young-wook Jun, Sassan Sheikholeslami, Daniel Hostetter, Cheryl Tajon, Charles Craik & A. Paul Alivisatos*
- “Continuous imaging of plasmon rulers in live cells reveals early stage caspase-3 activation at the single molecule level” Proceedings of National Academy of Sciences USA, 106, 17735 (2009).
- Haimei Zheng, Rachel K. Smith‡, Young-wook Jun‡, Christian Kisielowski, Ulrich Dahmen & A. Paul Alivisatos* “Observation of single colloidal platinum nanocrystal growth trajectories” Science 324, 1309 (2009).
- Jungwon Park, Haimei Zheng, Young-wook Jun & A. Paul Alivisatos* “Hetero-epitaxial anion exchange yields single-crystalline hollow nanoparticles” Journal of the American Chemical Society, 131, 13943-13945 (2009).
- Young-wook Jun, Jae-Hyun Lee, Mi-yun Kim & Jinwoo Cheon* “Chemical design of nanoparticle probes for ultra-sensitive magnetic resonance imaging” Angewandte Chemie International Edition, 47, 5122-5135 (2008).
(Selected as a frontispiece)
- Young-wook Jun, Jung-wook Seo & Jinwoo Cheon* “Nanoscaling laws of magnetic nanoparticles and their applicabilities in biomedical sciences” Accounts of Chemical Research, 41, 179-189 (2008).
- Jae-Hyun Lee, Yong-Min Huh, Young-wook Jun, Jung-wook Seo, Jung-tak Jang, Ho-taek Song, Sungjun Kim, Jin-Suck Suh & Jinwoo Cheon* “Artificially engineered magnetic nanoparticles for ultra-sensitive molecular imaging” Nature Medicine 13, 95-99 (2007).
- Jung-wook Seo, Young-wook Jun, Seung-won Park, Taeho Moon, Byungwoo Park & Jinwoo Cheon* “Two-dimensional nanosheet crystals” Angewandte Chemie International Edition, 46, 8828-8831 (2007).
- Young-wook Jun, Jin-sil Choi & Jinwoo Cheon* “Heterostructured magnetic nanoparticles: Their versatility and high performance capabilities” Chemical Communications 1203-1214 (2007).
- Jong-Il Park, Young-wook Jun, Jin-sil Choi & Jinwoo Cheon* “Highly crystalline anisotropic supercrystals via magnetic field induced assembly” Chemical Communications, 5001-5003 (2007).
- Yong-Min Huh, Eun-Suk Lee, Jae-Hyun Lee, Young-wook Jun, Pyung-Hwan Kim, Chae-Ok Yun, Joo-hang Kim, Jin-Suck Suh* & Jinwoo Cheon* “Hybrid nanoparticles for magnetic resonance imaging of target-specific viral gene delivery” Advanced Materials 19, 3109-3112 (2007). (Selected as an inside cover page)
- Young-wook Jun, Jung-tak Jang & Jinwoo Cheon* “Magnetic nanoparticle assisted molecular MR imaging” Bio-appl. Nanoparticles, 620, 82-106 (2007).
- Jinwoo Cheon, Jong-Il Park, Jin-sil Choi, Young-wook Jun, Sehun Kim, Min Gyu Kim, Young-Min Kim & Yoon Joong Kim “Magnetic superlattices and their nanoscale phase transition effects” Proceedings of National Academy of Sciences USA 103, 3023-3027 (2006).
- Young-wook Jun, Jin-sil Choi & Jinwoo Cheon* “Shape control of semiconductor and metal oxide nanocrystals through nonhydrolytic colloidal routes” Angewandte Chemie International Edition 45, 3414-3439 (2006).
(Selected as a frontispiece)
- Maria F. Casula, Young-wook Jun, David J. Zaziski, Anna Corrias & A. Paul Alivisatos* “The concept of delayed nucleation in nanocrystal growth: The case of iron oxide nanodisks” Journal of the American Chemical Society 128, 1675-1682 (2006).
- Jin-sil Choi, Young-wook Jun, Soo-In Yeon, Hyung Chan Kim, Jeon-Soo Shin* & Jinwoo Cheon* “Biocompatible heterostructured nanoparticles for multimodal biological detection” Journal of the American Chemical Society 128, 15982-15983 (2006).
- Jae-Hyun Lee, Young-wook Jun, Soo-In Yeon, Jeon-Soo Shin* & Jinwoo Cheon* “Dual mode nanoparticle probes for high performance magnetic resonance and fluorescence imaging of neuroblastoma” Angewandte Chemie International Edition 45, 8160-8162 (2006).
- Young-wook Jun,Jung-tak Jang & Jinwoo Cheon* “Nanocrystals and their biomedical applications” The Bulletin of Korean Chemical Society 27, 1-11 (2006).
- Young-wook Jun, Yong-Min Huh, Jin-sil Choi, Jae-Hyun Lee, Ho-Taek Song, Sungjun Kim, Sarah Yoon, Kyung-Sup Kim, Jeon-Soo Shin, Jin-Suck Suh* & Jinwoo Cheon* “Nanoscale size effect of magnetic nanocrystals and their utilization for cancer diagnosis via magnetic resonance imaging” Journal of the American Chemical Society 127, 5732-5733 (2005).
- Yong-Min Huh, Young-wook Jun,Ho-Taek Song, Sungjun Kim, Jin-sil Choi, Jae-Hyun Lee, Sarah Yoon, Kyung-Sup Kim, Jeon-Soo Shin, Jin-Suck Suh* & Jinwoo Cheon* “In vivo magnetic resonance detection of cancer by using magnetic nanocrystals” Journal of the American Chemical Society 127, 12387-12391 (2005).
- Ho-Taek Song, Jin-sil Choi, Yong-Min Huh, Sungjun Kim, Young-wook Jun, Jin-Suck Suh* & Jinwoo Cheon* “Surface modulation of magnetic nanocrystals in the development of highly efficient magnetic resonance probes for intracellular labeling” Journal of the American Chemical Society 127, 9992-9993 (2005).
- Young-wook Jun, Jae-Hyun Lee, Jin-sil Choi & Jinwoo Cheon* “Symmetry-controlled colloidal nanocrystals: Nonhydrolytic chemical synthesis and their shape determining parameters” Journal of Physical Chemistry B 109, 14795-14806 (2005). (Selected as a cover page)
- Jung-wook Seo, Young-wook Jun, Seung Jin Ko & Jinwoo Cheon* “In-situ one-pot synthesis of 1-dimensional transition metal oxide nanocrystals” Journal of Physical Chemistry B 109, 5389-5391 (2005).
- Young-wook Jun, Jung-wook Seo, Sang Joon Oh & Jinwoo Cheon* “Recent advances in the shape control of inorganic nano-building blocks” Coordination Chemistry Reviews 249, 1766-1775 (2005).
- Insik In, Young-wook Jun, Yoon-Joon Kim, Myung-Eun Seo & Sang Youl Kim* “Spontaneous one dimensional arrangement of spherical Au nanoparticles with liquid crystal ligands ” Chemical Communications, 800-801 (2005).
- Jong-Il Park, Min-Gyu Kim, Young-wook Jun, Jae-Sung Lee, Woo-Ram Lee & Jinwoo Cheon* “Characterization of superparamagnetic core-shell nanoparticles and monitoring their anisotropic phase transition to ferromagnetic ''solid solution'' nanoalloys” Journal of the American Chemical Society 126, 9072-9078 (2004).
- Insik In, Young-wook Jun & Sang Youl Kim* “Thiacrownether mediated size-controlled assembly of gold nanoparticles” Chemistry Letters 33, 1530-1531 (2004).
- Young-wook Jun, Maria F. Casula, Jae-Hwan Sim, Sang Youl Kim*, Jinwoo Cheon* & A. Paul Alivisatos* “Surfactant-assisted elimination of a high energy facet as a means of controlling the shapes of TiO2 nanocrystals” Journal of the American Chemical Society 125, 15981-15985 (2003).
- Young-wook Jun, Yun-Young Jung & Jinwoo Cheon* “Architectural control of magnetic semiconductor nanocrystals” Journal of the American Chemical Society 124, 615-619 (2002).
- Yong-Ho Kim, Young-wook Jun, Byung-Ho Jun, Sang-Min Lee & Jinwoo Cheon* “Sterically induced shape- and crystalline phase- control of gallium phosphide nanocrystals” Journal of the American Chemical Society 124, 13656-13657 (2002).
- Sang-Min Lee, Young-wook Jun,Sung-Nam Cho & Jinwoo Cheon* “Single crystalline star-shaped nanocrystals and their evolution: Programming the geometry of nano-building blocks” Journal of the American Chemical Society 124, 11244-11245 (2002).
- Jong-Il Park, Nam-Jung Kang, Young-wook Jun, Sang Jun Oh, Hyung Chan Ri & Jinwoo Cheon* “Superlattice and magnetism directed by size and shape of nanocrystals” Chemphyschem 3, 543-547 (2002).
- Young-wook Jun, Sang-Min Lee, Nam-Jun Kang & Jinwoo Cheon* “Controlled synthesis of multi-armed CdS nanorod architectures using mono-surfactant system” Journal of the American Chemical Society 123, 5150-5151 (2001).
- Young-wook Jun, Chang-Sik Choi & Jinwoo Cheon* “Size and shape controlled synthesis of ZnTe Nanocrystals with quantum confinement effect” Chemical Communications 101-102 (2001).
- Hong-GyuKang, Young-wook Jun, Jong-Il Park, Kyung-Bok Lee & Jinwoo Cheon* “Synthesis of porous palladium nanoballs and nanowires” Chemistry of Materials 12, 3530-3532 (2000).
- Young-wook Jun, Ja-Eung Koo & Jinwoo Cheon* “One step synthesis of size tuned zinc selenide quantum dots via a temperature controlled molecular precursor approach” Chemical Communications 1243-1244 (2000).
Book Chapters
- Young-wook Jun, Jae-Hyun Lee & Jinwoo Cheon* “Chapter 17. Nanoparticle contrast agents for molecular magnetic resonance imaging” in Nanobiotechnology II, Chad Mirkin & Christrof Niemeyer Eds. Wiley-VCH (2007).
- Young-wook Jun, Jung-tak Jang & Jinwoo Cheon* “Chapter 6. Magnetic resonance nanoparticle probes for cancer imaging” in Encyclopedia of Nanobiotechnology, Kumar Challa Ed. Wiley-VCH (2007).
- Young-wook Jun, Jung-tak Jang & Jinwoo Cheon* “Magnetic nanoparticle assisted molecular MR imaging” in Biological Applications of Engineered Nanostructures, Warren Chan Ed. Landes Sci. (2007).Young-wook Jun, Seung Jin Ko & Jinwoo Cheon* “Chapter 2. Colloidal inorganic nanocrystal building blocks” in Nanoparticle Assemblies and Superstructures, Nicholas Kotov Ed. CRC Press LLC. Aug. (2005).
- Jinwoo Cheon, Young-wook Jun & Sang-Min Lee “Chapter 4. Architecture of nanocrystal building blocks” in Nanoparticles: Building Blocks for Nanotechnology, Vincent Rotello Ed. Plenum Publishing Corp. (2003).
Research Team
- Jason Fabbri, PhD
Postdoc - Thomas Haas, MS
Staff Research Associate - Hyun Jung Lee, PhD
Postdoc - Daeha Seo, PhD
HFSP Postdoctoral Fellow - Kade Southard, BS
CCB Graudate Student
Meet our Team

Young-Wook Jun, PhD
Contact Us
Helen Diller Family Comprehensive Cancer Center
1450 3rd St. HD 365
San Francisco, CA 94158
United States
